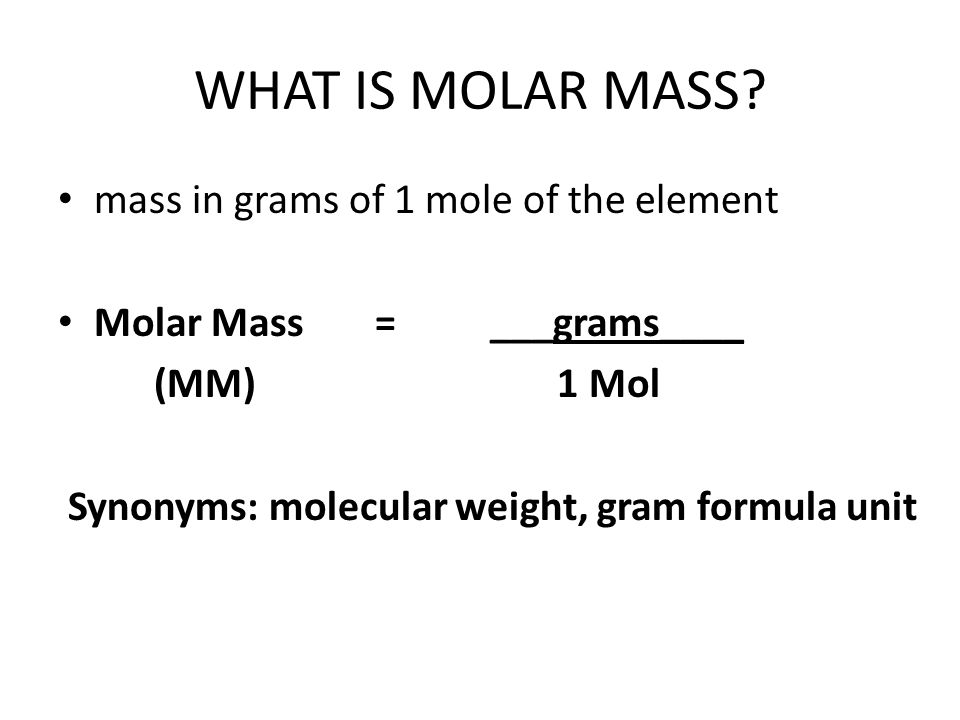PLEASE HELP QUICKLY!!! MM H2O2 = 34.02 g/mol MM H2O = 18.02 g/mol MM O2 = 32 g/mol 2H2O2 —> 2H2O + - Brainly.com

Bahco Wrench 9029C BH9029C Adjustable MOL Central Gran AP Wrench, Silver/Black, 6 Inch, 32 mm - - Amazon.com

SOLVED: What is the concentration in molarity of a solution which is 11.39 %m/v octane (MM = 114.2 g/mol ) in hexane (MM = 86.18 g/mol)?

Comparison of molecular mechanics-Poisson-Boltzmann surface area (MM-PBSA) and molecular mechanics-three-dimensional reference interaction site model ( MM-3D-RISM) method to calculate the binding free energy of protein-ligand complexes: Effect of metal ...

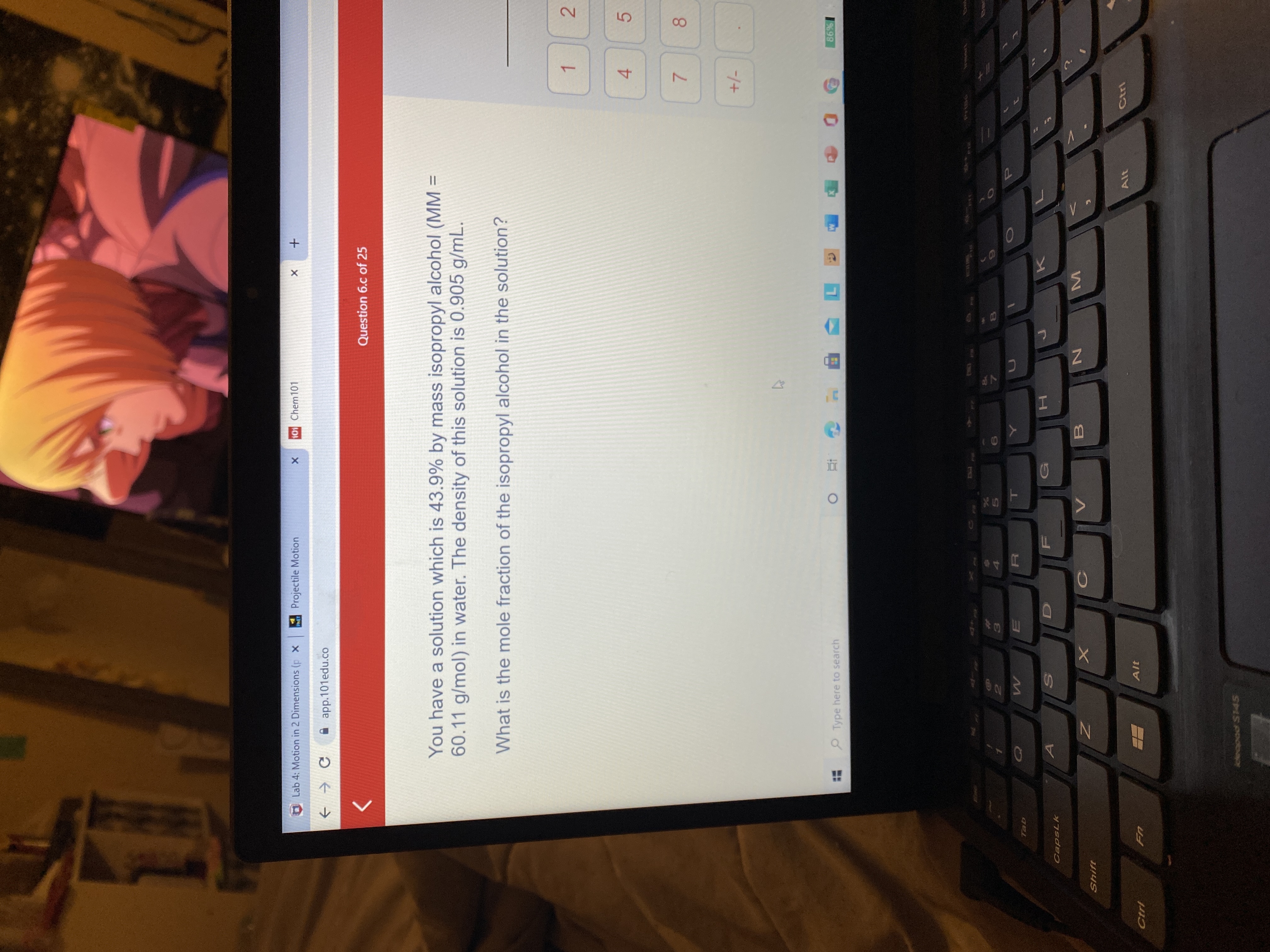


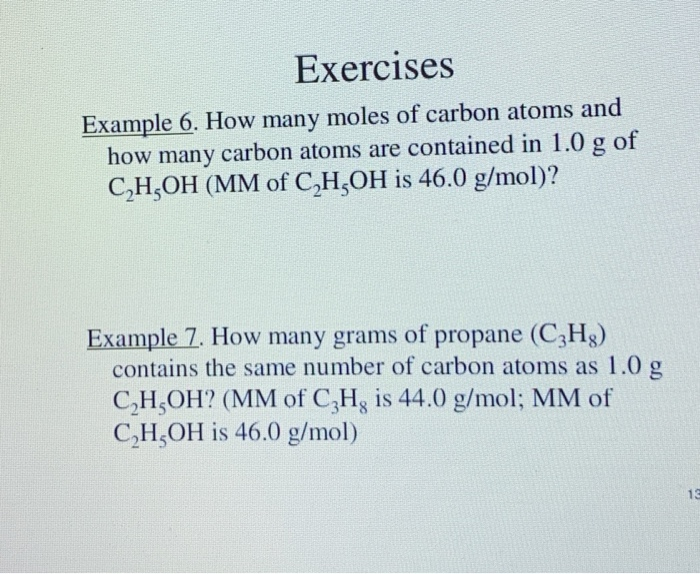
![ANSWERED] A solution is made using 180.3 g of hexan... - Physical Chemistry ANSWERED] A solution is made using 180.3 g of hexan... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/55090607-1659271777.3586252.jpeg)