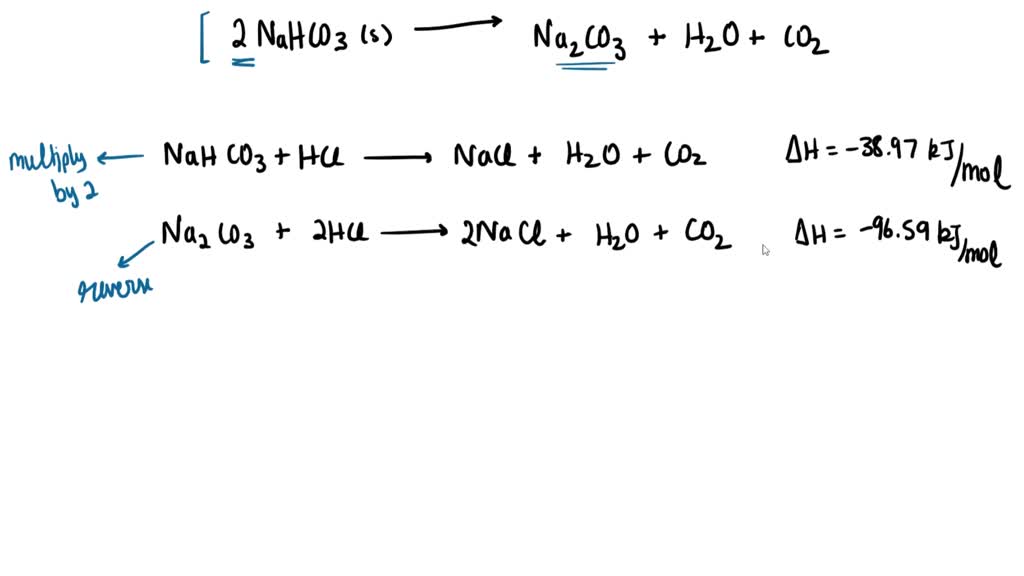
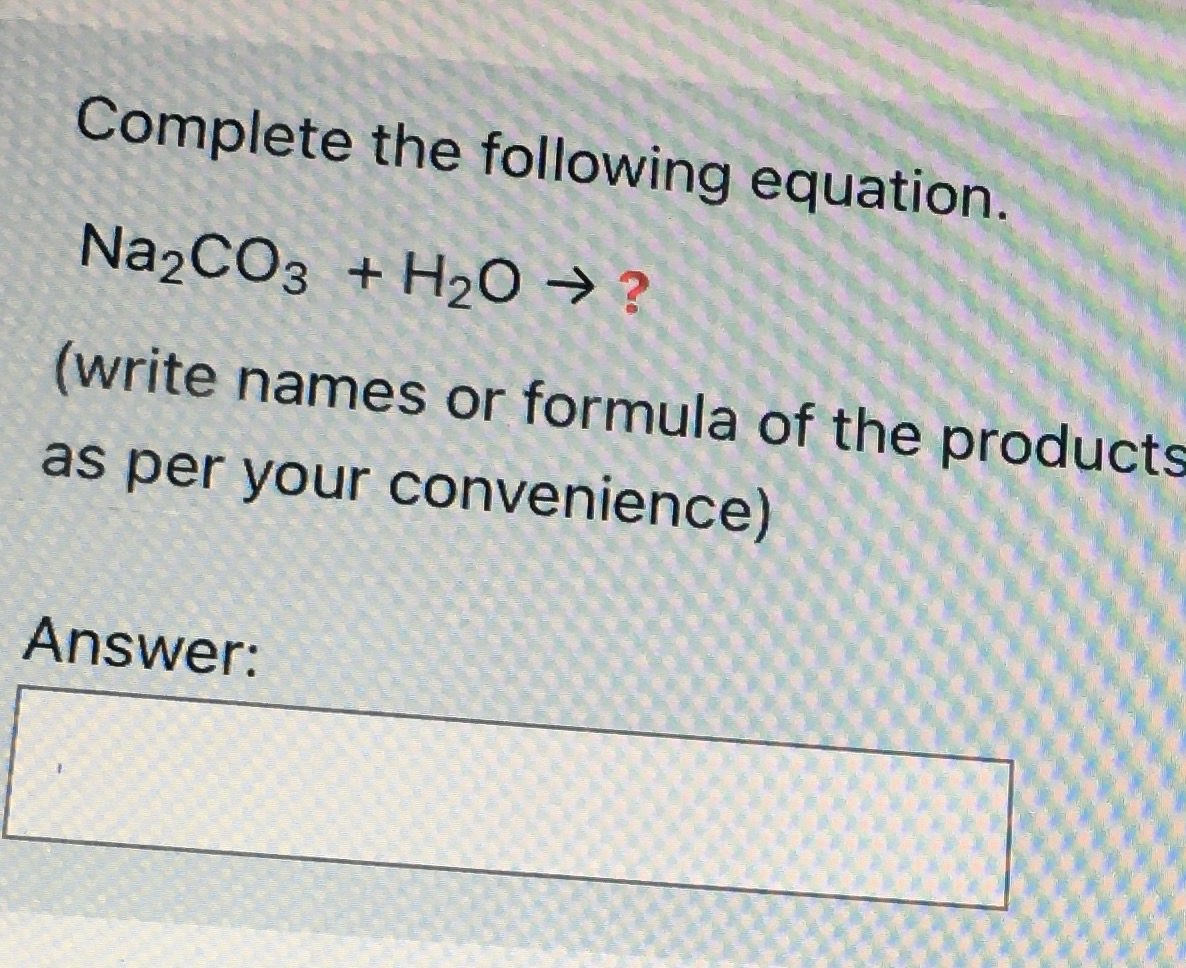
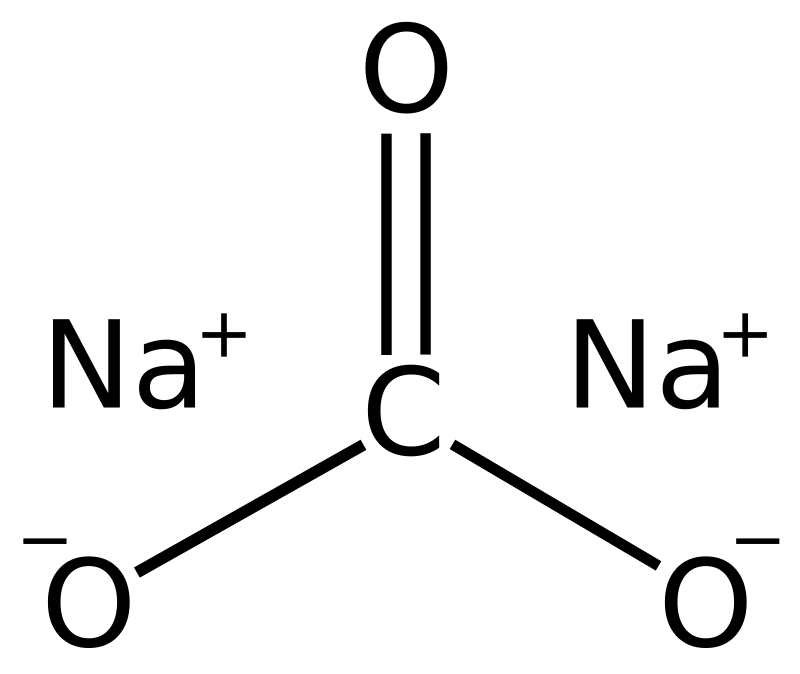
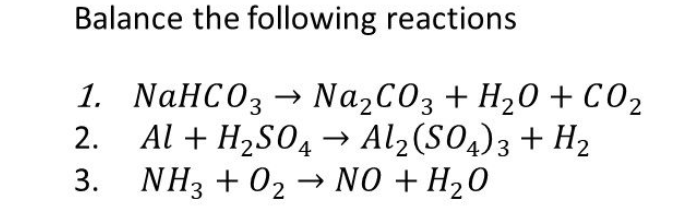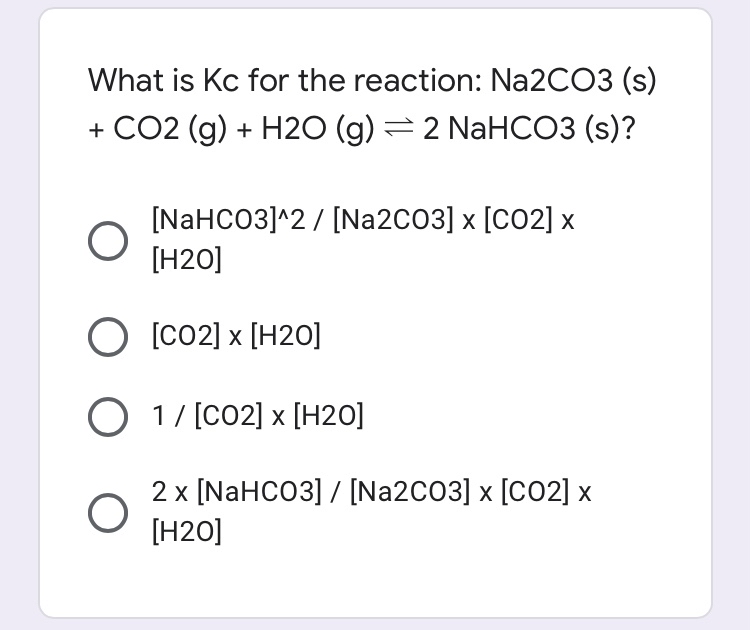Phase Equilibria of the NaOH–NaBO2–Na2CO3–H2O System at 30 °C, 60 °C, and 100 °C | Journal of Chemical & Engineering Data

5968-11-6, F.W. 124.00, Sodium Carbonate, Monohydrate, Granular, Reagent, ACS - 39G888|S1230-2.5KG76 - Grainger

Phase Equilibria of the NaOH–NaBO2–Na2CO3–H2O System at 30 °C, 60 °C, and 100 °C | Journal of Chemical & Engineering Data
Site-selective Suzuki–Miyaura coupling of heteroaryl halides – understanding the trends for pharmaceutically import