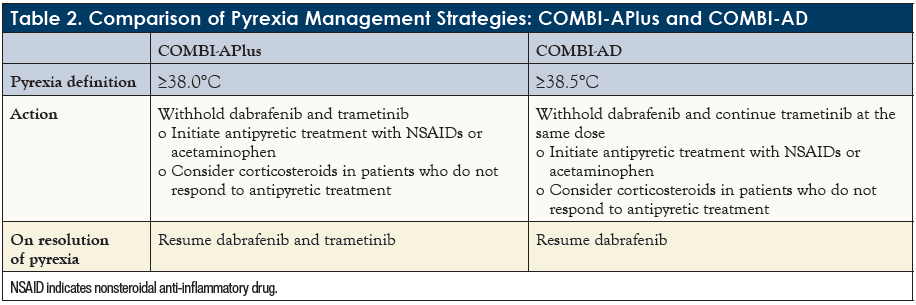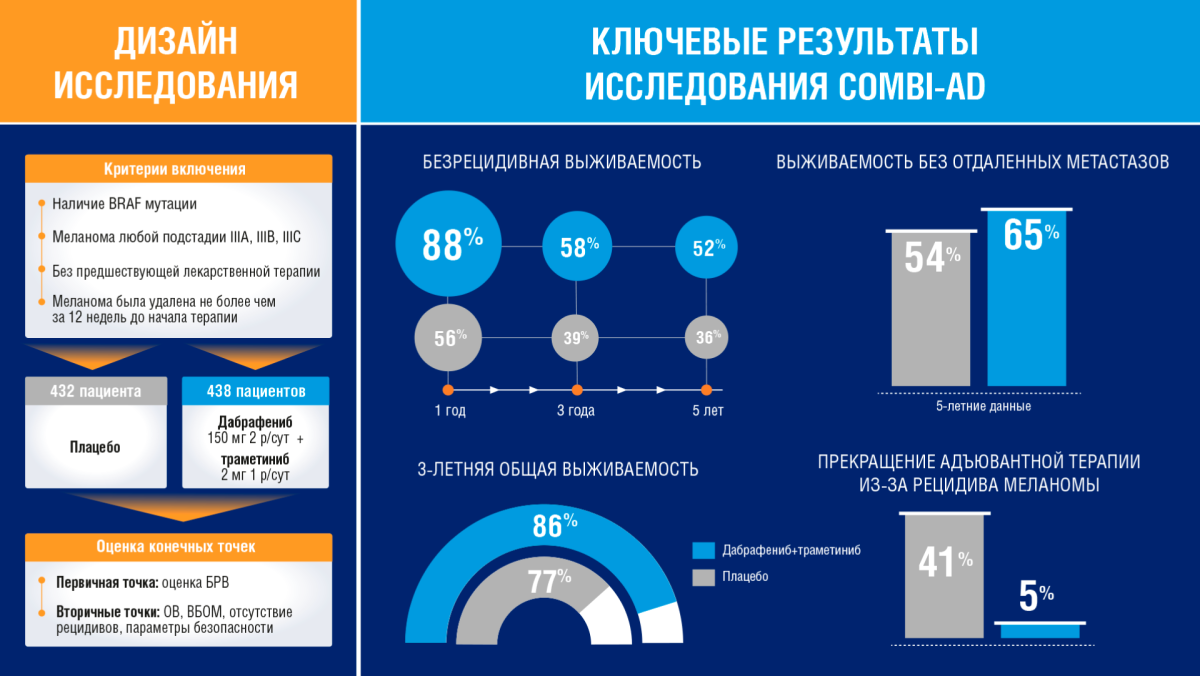
Исследование COMBI-AD. Адъювантная терапия дабрафенибом и траметинибом при меланоме III стадии с мутацией BRAF | meducate.ru

Sayed ali on Twitter: "Updated adj D and T COMBI AD .29% patient discontinued treatment. #WMC2017 #melanoma #melanoma2017 https://t.co/kbx3i5Esgm" / Twitter

Please note, these are the actual video-recorded proceedings from the live CME event and may include the use of trade names and other raw, unedited content. - ppt download

Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAFV600-mutant, stage III melanoma (COMBI-AD): exploratory biomarker analyses from a randomised, phase 3 trial - The Lancet Oncology

ESMO 2017: COMBI-AD: Adjuvant Dabrafenib (D) Plus Trametinib (T) for Resected Stage III BRAF V600E/K–Mutant Melanoma

Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAFV600-mutant, stage III melanoma (COMBI-AD): exploratory biomarker analyses from a randomised, phase 3 trial - The Lancet Oncology